This is also apparent in reports of produced water re-injection. We can conclude that low-salinity waterflooding provides adequate conditions for formation of relatively stable emulsions, provided kaolinite detachment occurs. Salinity, especially calcium concentration, affects the morphology and stability of emulsions. The first work involved the Pickering emulsion with Hal is published by. Zeta potential measurements and bottle tests clearly show that clay particles with zero point of charge (ZPC) at low pH become less effective at stabilizing emulsions in contrast with reservoir conditions, while silica shows the opposite behavior, stabilizing better emulsion at ZPC. In contrast to surfactant molecules, which are continually adsorbed and desorbed, in the case of Pickering emulsions, the particles are adsorbed at the. The reason may be the changed surface charges of Hal in varied pH or salt solution. Also, kaolinite of the order of 1μm in size more easily adsorb at the oil-water interface, while silica with larger diameter, such as 5μm, can form more stable emulsions than with 1μm and 3μm. No stable emulsion forms for the methyl analogue, but very stable o/w emulsions can be prepared with the other three members, with the minimum concentration of salt being required decreasing with R chain length to as low as 5 × 10 -5 M. Our results show that both kaolinite and silica promote emulsion stability. 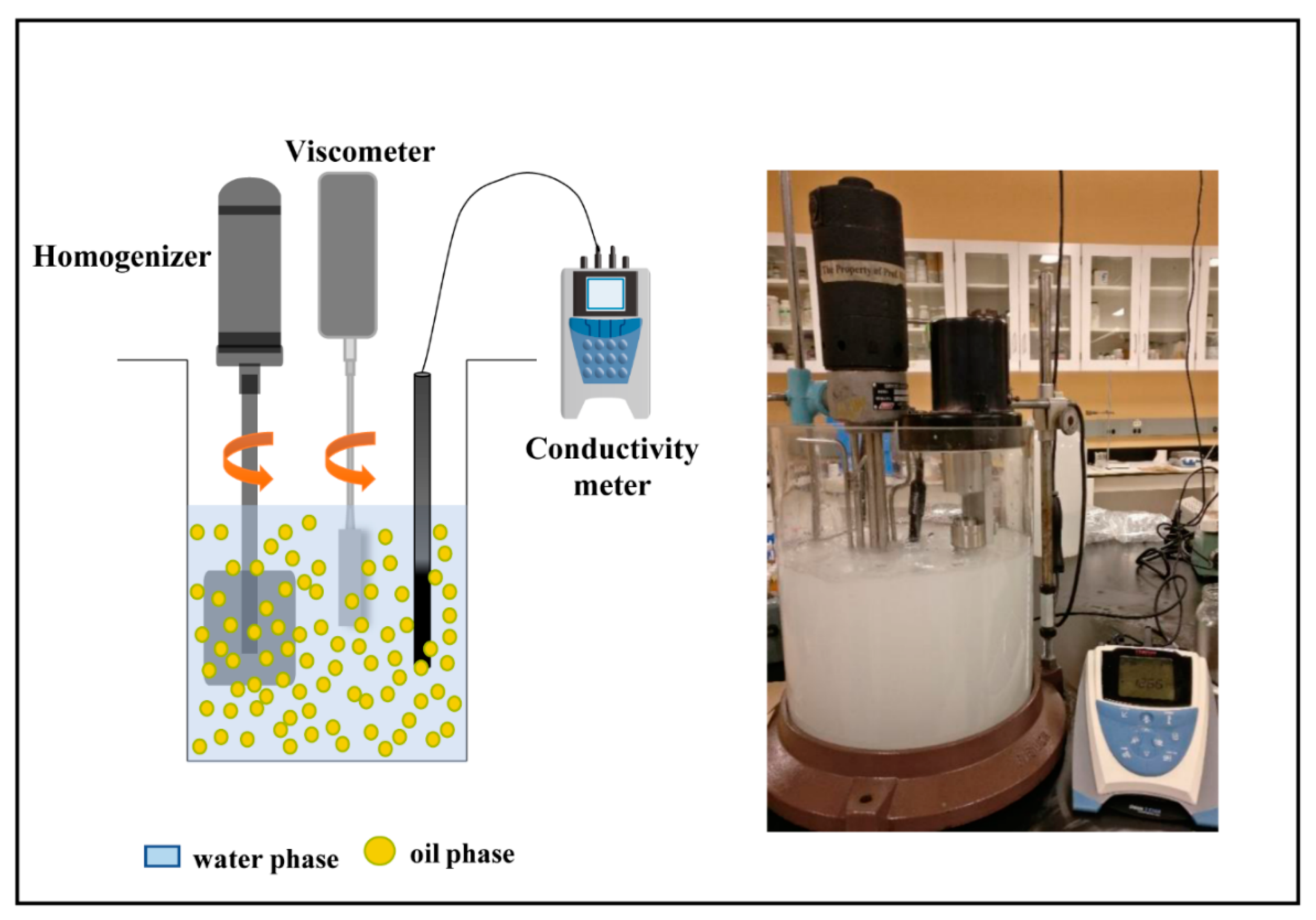
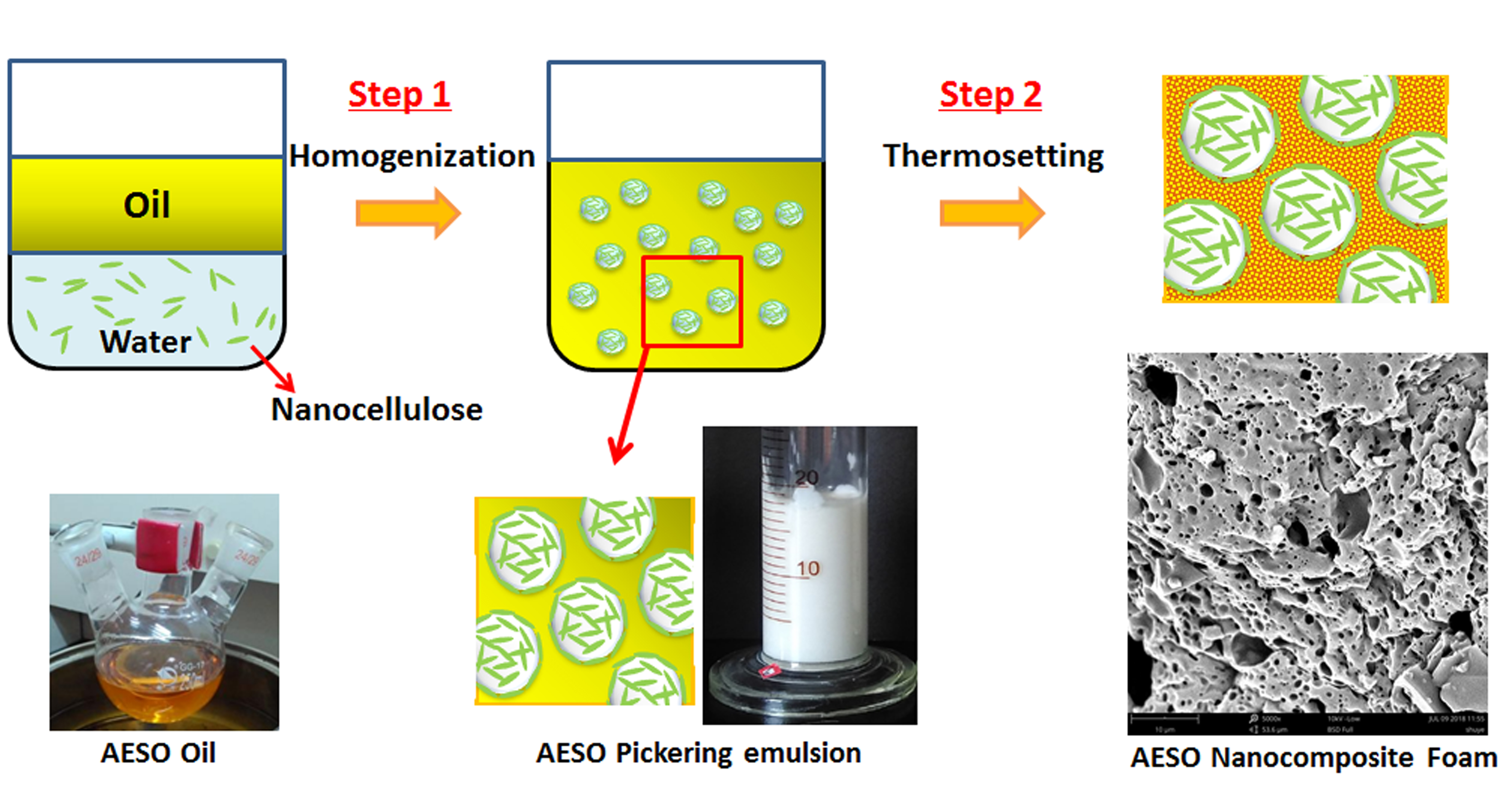
1 Different from the thermodynamically stable micro-emulsions, of which the droplets are smaller than 100 nm, macro-emulsions are thermodynamically unstable and have larger droplets in the range of 0.110 m. The stability of emulsion was determined by simple bottle tests and by using an optical microscope equipped with a digital camera. 1 Introduction Macro-emulsions are mixtures of two immiscible liquids (usually water and an organic solvent). Clay particle-size distribution, which impacts the formation and stability of emulsions, was obtained using light diffraction. The clay zeta potential at different pH was measured to estimate the effect of particle charge on emulsion stability. Also, the emulsion was prepared by mixing this oil with 100 times dilution dispersed with particles to investigate pH effect on emulsion stability. Once the emulsion has formed, salt has an influence on the final position of trapped particles as described by, w, the three-phase contact angle. Examples of Pickering emulsions include dry water, a water-air emulsion. First, synthetic reservoir brine with 1, 10 and 100 times dilution was emulsified with crude oils of commercial interest, at several water-oil ratios, on which salinity effect on emulsion stability was studied. experiments showed that CNC should be combined with salt in order to. The stability and morphology of crude-oil emulsions with and without suspended kaolinite or silica were investigated as functions of salinity. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
